Photochemistry is the discipline that studies the chemical processes in which light plays a role. In this post, we do not aim to carry out a thorough analysis about photochemistry, but to provide a brief overview about some concepts we find interesting in this field. In the first place, we are going to describe the basic operation of a solar simulator. Then, we will explain two different phenomena: photodegradation, and photocatalysis.
Solar simulator
A solar simulator is a lighting system that tries to reproduce, as closely as possible, the characteristics of natural sunlight. There are different technical standards to establish the performance of a solar simulator, but they always consider the following three different parameters:
• Spectral distribution: match to the standard spectrum.
• Spatial uniformity: distribution and consistency of irradiance over an area.
•Temporal stability: consistency of irradiance over time.
Each one of the previous parameters can be evaluated with an A, B, or C, being A the best class, and C the worst class (if the specifications of the solar simulator are worse than class C, it is classified as class U, which stands for unclassified).
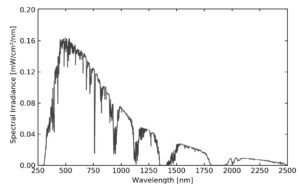
Regarding the spectral distribution, different standards can be employed. A standard establishes the percentage of the total irradiance that has to be emitted in a certain wavelength range. The most common standard is AM1.5G (see Figure 1). The letters ‘AM’ mean ‘air mass’, that is, the spectrum of light that has travelled through air. ‘1.5’ corresponds to a zenith angle of 48.2°, representative value for mid-latitudes. Finally, ‘G’ means global, indicating that the spectrum also considers diffuse and scattered light. It is also worth mentioning that the standard states that the total irradiance of an AM1.5G simulator has to be 1000 W/m2, which is called “a sun”.
There are two types of solar simulators: continuous or pulsed. Here, different type of light sources can be used with that purpose, including metal halide arc lamps, quartz-tungsten halogen (QTH) lamps, xenon arc lamps and LED light sources, among others. LED solar simulators offer the best temporal stability, low consumption, long lifetime and precise control. Contact us through info@pyroistech.com if you are interested in a LED solar simulator.
Photodegradation
Photodegradation consists in the alteration of materials by light, usually from wavelengths in the ultraviolet (UV) (250 – 400 nm) or visible (400 – 700 nm) part of the spectrum. In particular, many studies are focused on this phenomenon in the case of polymers, as UV light can break the polymer chains and cause adverse effects such as cracking, chalking, color changes, and the loss of physical properties. The term photo-oxidation is employed when the degradation caused by light is helped by the presence of oxidants. In addition to this, photo-oxidation can also be accelerated by thermal degradation.
Although there are some polymers (for instance, PET) that exhibit a moderate resistance to the effects of UV light, there are other widely employed polymers that are severely affected, including polystyrene (PS), polyvinyl chloride (PVC), polypropylene (PP), or polyethylene (PE). In order to protect these materials, UV stabilizers are added. UV stabilizers consist of chemical components that absorb UV light while reducing the photodegradation of the polymer they are added in.
Photodegradation in polymers is studied by researchers by means of a solar simulator (described in the previous point), as the UV part of sunlight is the most common source of photodegradation in real life applications, or by directly using UV sources, such as UV LED sources. You can check our range of UV LED sources in this link.
Photocatalysis
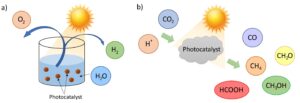
Photocatalysis can be defined as the acceleration of a photoreaction (reaction that involves or requires light) by the presence of a catalyst. Two classical photocatalytic reactions include water splitting and CO2 reduction (see Figure 2).
Water splitting consists in producing hydrogen and oxygen from water, thus it is considered an attractive and clean technology to generate hydrogen, which is usually obtained from petroleum and natural gas. The interest in hydrogen arises from the fact that it can be used as a way of storing renewable energy. Particularly, titanium dioxide (TiO2), which works under UV light, has been typically used as photo-catalyst in water splitting reactions, although nowadays other catalysts are being explored.
Regarding CO2, the concern about climate change has led scientists to search for strategies to lower its emissions or even capture it from the atmosphere. The advantage of the photocatalytic reduction of CO2 over other approaches is that it can be carried out at lower temperatures and pressures, thus decreasing energy consumption. Materials that are employed as catalysts in this case include TiO2, g-C3N4 (graphitic carbon nitride) and perovskites. UV and visible sources are usually employed in this photocatalytic reaction. See our UV and visible LED light sources in our catalogue on this link.
Written by J.J. Imas
Bibliography
[1] Solar simulation technology. G2V.
[2] Solar simulator. Wikipedia
[3] Photo-oxidation of polymers. Wikipedia
[4] Photodegradation and stabilization of polymers. EPFL École polytechnique fédérale de Lausanne.
[5] Photodegradation. Science Direct. Elsevier
